
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 1 of 20
Statistical Analysis Plan
Version 1.0
Date: 09 June 2020
Protocol version: 6.0, 14 May 2020
IRAS no: 281712
REC ref: EE/20/0101
ISRCTN: 50189673
EudraCT: 2020-001113-21
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 2 of 20
Table of Contents
Abbreviations ......................................................................................................................... 4
List of authors and reviewers................................................................................................. 5
Roles and responsibilities ...................................................................................................... 6
1 Introduction ....................................................................................................................... 7
2 Background information .................................................................................................... 7
2.1 Rationale ..................................................................................................................... 7
2.2 Objectives of the trial .................................................................................................. 7
Primary objective ................................................................................................. 7
Secondary objectives ........................................................................................... 7
2.3 Trial design .................................................................................................................. 7
2.4 Eligibility ...................................................................................................................... 8
Inclusion criteria................................................................................................... 8
Exclusion criteria .................................................................................................. 8
2.5 Treatments .................................................................................................................. 8
Main randomisation part A: ................................................................................. 8
Main randomisation part B: ................................................................................. 8
Second randomisation for patients with progressive COVID-19 ......................... 9
2.6 Definitions of primary and secondary outcomes ........................................................ 9
Primary outcome ................................................................................................. 9
Secondary clinical outcomes ................................................................................ 9
Subsidiary clinical outcomes ................................................................................ 9
Detailed derivation of outcomes ....................................................................... 10
2.9 Randomisation .......................................................................................................... 10
Main randomisation part A ................................................................................ 10
Main randomisation part B ................................................................................ 11
Second randomisation for patients with progressive COVID-19 ....................... 11
2.10 Blinding .................................................................................................................. 11
2.11 Data collection schedule........................................................................................ 11
2.12 Data monitoring ..................................................................................................... 12
2.13 Trial reporting ........................................................................................................ 12
3 Analysis populations ........................................................................................................ 12
3.1 Population definitions ............................................................................................... 12
4 Descriptive analyses ......................................................................................................... 12
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 3 of 20
4.1 Participant throughput .............................................................................................. 12
4.2 Baseline comparability of randomised groups ......................................................... 12
Main randomisation (part A and B) ................................................................... 13
Second randomisation ....................................................................................... 13
4.3 Completeness of follow-up ....................................................................................... 13
4.4 Adherence to treatment ........................................................................................... 13
5 Comparative analyses ...................................................................................................... 14
5.1 Main randomisation part A ....................................................................................... 14
Primary outcome ............................................................................................... 14
Secondary outcomes .......................................................................................... 14
5.1.2.1 Time to discharge from hospital .................................................................... 14
5.1.2.2 Use of mechanical ventilation/ECMO or death (among those not on
ventilation or ECMO at randomisation) ........................................................................... 14
Subsidiary clinical outcomes .............................................................................. 15
5.1.3.1 Cause-specific mortality ................................................................................. 15
5.1.3.2 Use of renal dialysis or haemofiltration ......................................................... 15
5.1.3.3 Major cardiac arrhythmia............................................................................... 15
5.1.3.4 Use of ventilation (overall and by type) ......................................................... 15
5.1.3.5 Duration of ventilation (overall and by type) ................................................. 15
5.2 Main randomisation part B ....................................................................................... 15
5.3 Second randomisation .............................................................................................. 16
5.4 Pre-specified subgroup analyses ............................................................................... 16
5.5 Significance levels and adjustment of p-values for multiplicity ................................ 16
5.6 Statistical software employed ................................................................................... 17
5.7 Data standards and coding terminology ................................................................... 17
6 Safety data ....................................................................................................................... 17
7 Additional exploratory analysis ....................................................................................... 17
8 DIFFERENCES FROM protocol V6.0 .................................................................................. 17
9 References ....................................................................................................................... 17
9.1 Trial documents ......................................................................................................... 17
9.2 Other references ....................................................................................................... 17
10 Approval ........................................................................................................................... 19
11 Document history ............................................................................................................ 20
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 4 of 20
Abbreviations
ADaM
Analysis Data Model
AE
adverse event
CDISC
The Clinical Data Interchange Standards Consortium
CI
confidence interval
CoV
Coronavirus
COVID
coronavirus-induced disease
CPAP
Continuous Positive Airway Pressure
CRP
C-reactive protein
CTU
clinical trials unit
CTSU
Clinical Trials Service Unit
DMC
Data Monitoring Committee
ECMO
Extra Corporeal Membrane Oxygenation
eCRF
Electronic case report form
FiO
2
fraction of inspired oxygen
ICD
International Classification of Diseases
IFN
interferon
ICNARC
Intensive Care National Audit and Research Centre
IQR
interquartile range
ITT
intention to treat
MedDRA
Medical Dictionary for Regulatory Activities
MERS
Middle East Respiratory Syndrome
NPEU
National Perinatal Epidemiology Unit
OPCS-4
NHS Classification of Interventions and Procedures
PaO
2
partial pressure of oxygen
RR
risk ratio
SAE
serious adverse event
SARS
severe acute respiratory syndrome
SARS-CoV-2
virus causing COVID-19
SSAR
Suspected serious adverse reaction
SUSAR
Suspected unexpected serious adverse reaction
SD
standard deviation
SC
Steering Committee
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 5 of 20
List of authors and reviewers
Authors
Dr Louise Linsell, Lead Trial Statistician, Nuffield Department of Population Health (NDPH),
University of Oxford
Jennifer Bell, Trial Statistician, NDPH, University of Oxford
Reviewers
Professor Jonathan Emberson, Data Monitoring Committee (DMC) Statistician, NDPH,
University of Oxford (prior to unblinded interim analysis of trial outcomes)
Professor Richard Haynes, Steering Committee (SC) Member, NDPH, University of Oxford
Professor Peter Horby, Chief Investigator (CI), Epidemic Diseases Research Group, University
of Oxford
Professor Thomas Jaki, Steering Committee Member, Department of Mathematics and
Statistics, Lancaster University
Associate Professor Edmund Juszczak, SC Member, NDPH, University of Oxford
Professor Martin Landray, Deputy CI, NDPH, University of Oxford
Professor Alan Montgomery, SC Member, Nottingham Clinical Trials Unit, University of
Nottingham
Dr Natalie Staplin, DMC Statistician, NDPH, University of Oxford (prior to unblinded interim
analysis of trial outcomes)
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 6 of 20
Roles and responsibilities
Trial Statisticians
Dr Louise Linsell and Jennifer Bell
NDPH, University of Oxford
Role: To develop the statistical analysis plan and conduct the final comparative analysis.
Blinded to trial allocation.
Data Monitoring Committee (DMC) Statisticians
Professor Jonathan Emberson and Dr Natalie Staplin
NDPH, University of Oxford
Role: To conduct regular interim analyses for the DMC. Contribution restricted up until
unblinded to trial allocation.
Trial IT systems & Programmers
Andy King, David Murray, Richard Welsh
NDPH, University of Oxford
Role: To generate and prepare reports monitoring the randomisation schedule. To supply
data snapshots for interim and final analysis. Responsibility for randomisation system, clinical
databases and related activities.
Bob Goodenough
NDPH, University of Oxford
Role: Validation of IT systems
Dr Will Stevens
NDPH, University of Oxford
Role: To produce analysis-ready datasets according to CDISC standards.

RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 7 of 20
1 INTRODUCTION
This document details the proposed presentation and analysis for the main paper(s) reporting
results from the multicentre randomised controlled trial RECOVERY (ISRCTN50189673) to
investigate multiple treatments on major outcomes in inpatients for COVID-19 (clinically
suspected or laboratory confirmed).
The results reported in these papers will follow the strategy set out here, which adheres to
the guidelines for the content of a statistical analysis plan.
1
Any subsequent analyses of a
more exploratory nature will not be bound by this strategy, and will be detailed in a separate
analysis plan.
Suggestions for subsequent analyses by oversight committees, journal editors or referees, will
be considered carefully in line with the principles of this analysis plan.
Any deviations from the statistical analysis plan will be described and justified in the final
report to the funder. The analysis will be carried out by an identified, appropriately qualified
and experienced statisticians, who will ensure the integrity of the data during their processing
e.g. by parallel programming.
This statistical analysis plan is based on the latest version of the protocol. A record of
amendments to the protocol can be found in the RECOVERY trial directory:
https://www.recoverytrial.net/for-site-staff/site-set-up-1.
2 BACKGROUND INFORMATION
2.1 Rationale
In early 2020, as the protocol was being developed, there were no approved treatments for
COVID-19. The aim of the trial is to provide reliable evidence on the efficacy of candidate
therapies (including re-purposed and novel drugs) for suspected or confirmed COVID-19
infection on major outcomes in hospitalised adult patients receiving standard care.
2.2 Objectives of the trial
Primary objective
To provide reliable estimates of the effect of study treatments on all-cause mortality within
28 days of randomisation.
Secondary objectives
To investigate the effect of study treatments on the duration of hospital stay, the need for
(and duration of) ventilation, and the need for renal replacement therapy.
2.3 Trial design
This is a multi-centre, multi-arm, adaptive, open-label, randomised controlled trial with three
possible stages of randomisation. In the main randomisation patients are allocated to no
additional treatment or one of 4 anti-viral or host-directed treatments. In addition, in a

RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 8 of 20
factorial design, eligible patients can also be allocated simultaneously to no additional
treatment or convalescent plasma. Patients who deteriorate according to predefined criteria
can be further randomised to no additional treatment or an immunomodulatory treatment.
The trial is designed with streamlined processes in order to facilitate rapid large-scale
recruitment with minimal data collection.
2.4 Eligibility
Inclusion criteria
Patients are eligible for the trial if all of the following are true:
Hospitalised
SARS-Cov-2 infection (clinically suspected or laboratory confirmed)
No medical history that might, in the opinion of the attending clinician, put the patient
at significant risk if they were to participate in the trial.
Exclusion criteria
If one or more of the active drug treatments is not available at the hospital or is believed, by
the attending clinician, to be contraindicated (or definitely indicated) for the specific patient,
then this fact will be recorded via the web-based form prior to randomisation; random
allocation will then be between the remaining (or indicated) arms.
2.5 Treatments
All patients will receive standard management for the participating hospital. The main
randomisation will be between the following treatment arms (although not all arms may be
available at any one time). The doses listed are for adults; paediatric dosing is described in
the protocol.
Main randomisation part A:
No additional treatment
Lopinavir 400mg-Ritonavir 100mg by mouth (or nasogastric tube) every 12 hours for
10 days.
Corticosteroid in the form of dexamethasone, administered as an oral liquid or
intravenous preparation 6 mg once daily for 10 days. In pregnancy, prednisolone 40
mg administered by mouth (or intravenous hydrocortisone 80 mg twice daily) should
be used instead.
Hydroxychloroquine by mouth for 10 days (4 doses in first 24 hours and 1 dose every
12 hours for 9 days).
Azithromycin 500mg by mouth (or nasogastric tube) or intravenously once daily for a
total of 10 days.
Main randomisation part B:
In a factorial design, eligible patients may be randomised to the arms below. The doses listed
are for adults; paediatric dosing is described in the protocol.

RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 9 of 20
No additional treatment
Convalescent plasma Single unit of ABO compatible convalescent plasma (275mls +/-
75 mls) intravenous per day on study days 1 (as soon as possible after randomisation)
and 2 (with a minimum of 12 hour interval between 1st and 2nd units). ABO identical
plasma is preferred if available. The second transfusion should not be given if patient
has a suspected serious adverse reaction during or after the first transfusion.
Second randomisation for patients with progressive COVID-19
Patients enrolled in the main RECOVERY trial and with clinical evidence of a hyper-
inflammatory state may be considered for a second randomisation if they meet the following
criteria:
Randomised into the main RECOVERY trial no more than 21 days ago
Clinical evidence of progressive COVID-19:
- oxygen saturation <92% on room air or requiring oxygen (or in children, significant
systemic disease with persistent pyrexia, with or without evidence of respiratory
involvement); and
- C-reactive protein (CRP) ≥75 mg/L
No medical history that might, in the opinion of the attending clinician, put the patient
at significant risk if they were to participate in this aspect of the RECOVERY trial
Eligible participants may be randomised between the following treatment arms:
No additional treatment
Tocilizumab by intravenous infusion with the dose determined by body weight.
2.6 Definitions of primary and secondary outcomes
Outcomes will be assessed at 28 days and then 6 months after randomisation. Analysis of
longer-term outcomes collected beyond this will be described in a separate Statistical Analysis
Plan.
Primary outcome
Mortality (all-cause)
Secondary clinical outcomes
Time to discharge from hospital
Use of mechanical ventilation/Extra Corporal Membrane Oxygenation (ECMO) or
death (among patients not on ventilation or ECMO at baseline)
Subsidiary clinical outcomes
Cause-specific mortality (COVID-19; cardiovascular; non-vascular; other)
Use of renal dialysis or haemofiltration
Serious cardiac arrhythmia (recorded in a subset)

RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 10 of 20
Use of ventilation (overall and by type)
Duration of ventilation (overall and by type)
Detailed derivation of outcomes
The detailed derivation of outcomes included in statistical analysis will be described
separately in a data derivation document and included in the Study Data Reviewer’s Guide.
2.7 Hypothesis framework
For each of the primary, secondary and subsidiary outcomes, the null hypothesis will be that
there is no true difference in effect between any of the treatment arms.
2.8 Sample size
The larger the number randomised, the more accurate the results will be, but the numbers
that can be randomised will depend critically on how large the epidemic becomes. If
substantial numbers are hospitalised in the participating centres then it may be possible to
randomise several thousand with moderate disease and a few thousand with severe disease.
Some indicative sample sizes and projected recruitment will be estimated using emerging
data for several different scenarios. Sample size and recruitment will be monitored by the
Steering Committee (SC) throughout the trial.
2.9 Randomisation
Eligible patients will be randomised using a 24/7 secure central web-based randomisation
system, developed and hosted within NDPH, University of Oxford. Users of the system will
have no insight into the next allocation, given that simple randomisation is being used. In the
event that a patient is randomised inadvertently more than once during the same hospital
admission, the first allocation will be used.
The implementation of the randomisation procedure will be monitored by the Senior Trials
Programmer, and the SC notified if an error in the randomisation process is identified.
Main randomisation part A
Simple randomisation will be used with a 2:1:1:1:1 allocation ratio to one of the following
treatment arms (in addition to usual care), which is subject to change:
No additional treatment
Lopinavir-Ritonavir
Corticosteroid
Hydroxychloroquine
Azithromycin
The randomisation programme will allocate patients in a ratio of 2:1 between the no
additional treatment arm and each of the other arms that are not contra-indicated and
available. Hence if all 4 active treatment arms are available, then the randomisation will be in
the ratio 2:1:1:1:1. If one or more of the active drug treatments is not available at the hospital
or is believed, by the attending clinician, to be contraindicated (or definitely indicated) for the

RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 11 of 20
specific patient, then this fact will be recorded via the web-based form prior to randomisation;
random allocation will then be between the remaining arms (in a 2:1:1:1, 2:1:1 or 2:1 ratio).
Main randomisation part B
In a factorial design, eligible patients will be randomised simultaneously using simple
randomisation with allocation ratio 1:1 to one of the following arms:
No additional treatment
Convalescent plasma
Second randomisation for patients with progressive COVID-19
Eligible participants may be randomised using simple randomisation with an allocation ratio
1:1 between the following arms:
No additional treatment
Tocilizumab
2.10 Blinding
This is an open-label study. However, while the study is in progress, access to tabular results
of study outcomes by treatment allocation will not be available to the research team, CIs, trial
statisticians, clinical teams, or members of the SC (unless the DMC advises otherwise). The
DMC and DMC statisticians will be unblinded.
2.11 Data collection schedule
Baseline and outcome information will be collected on trial-specific electronic case report
forms (eCRFs) and entered into a web-based IT system by a member of the hospital or
research staff. Follow-up information will be collected on all study participants, irrespective
of whether or not they complete the scheduled course of allocated study treatment. Study
staff will seek follow-up information through various means, including routine healthcare
systems and registries.
All randomised participants will be followed up until death or 6 months post-randomisation
to the main trial (whichever is sooner). NHS Digital and equivalent organisations in the
devolved nations will supply data fields relevant to trial baseline and outcome measures to
NDPH, University of Oxford on a regular basis, for participants enrolled into the trial. This will
be combined with the trial-specific data collected via the web-based IT system and
adjudicated internally.
Longer term (up to 10 years) follow-up will be sought through linkage to electronic healthcare
records and medical databases including those held by NHS Digital, Public Health England and
equivalent bodies, and to relevant research databases (e.g. UK Biobank, Genomics England).
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 12 of 20
2.12 Data monitoring
During the study all study data will be supplied in strict confidence to the independent DMC
for independent assessment and evaluation. The DMC will request such analyses at a
frequency relevant to the emerging data from this and other studies.
The DMC has been requested to determine if, in their view, the randomised comparisons in
the study have provided evidence on mortality that is strong enough (with a range of
uncertainty around the results that is narrow enough) to affect national and global treatment
strategies. Hence, multiple reviews by the Data Monitoring Committee have no material
impact on the final analysis. In such a circumstance, the DMC will inform the SC who will make
the results available to the public and amend the trial arms accordingly.
2.13 Trial reporting
The trial will be reported according to the principles of the CONSORT statements.
2, 3, 4
The
exact composition of the trial publication(s) depends on the size of the epidemic, the
availability of drugs, and the findings from the various pairwise comparative analyses (with
the no additional treatment arm) in the main trial.
3 ANALYSIS POPULATIONS
3.1 Population definitions
The intention to treat (ITT) population will be all participants randomised, irrespective of
treatment received. This ITT population will be used for analysis of efficacy and safety data.
For interim analyses, baseline data will be reported for all participants with data available and
outcome data will be reported for all participants who have died, been discharged from
hospital, or reached day 28 after the first randomisation.
4 DESCRIPTIVE ANALYSES
4.1 Participant throughput
The flow of participants through the trial will be summarised for each separate pairwise
comparison using a CONSORT diagram, for the main and second randomisation separately.
The flow diagram for the nested factorial design (main randomisation part B to convalescent
plasma) will be stratified by the 5 arms included in the main randomisation part A. The flow
diagrams will describe the numbers of participants randomly allocated, who received
allocation, withdrew consent, and included in the ITT analysis population. The flow diagrams
for arms in the main randomisation will also report the number of participants who
underwent the second randomisation.
4.2 Baseline comparability of randomised groups
The following characteristics will be described separately for patients randomised to each
main comparison (for each separate pairwise comparison of active treatment with the no
additional treatment arm), and separately for the first and second randomisation.

RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 13 of 20
Main randomisation (part A and B)
Age at randomisation
Sex
Ethnicity
Time since COVID-19 symptoms onset
Time since hospitalisation
Current respiratory support requirement
Currently requiring renal dialysis or haemofiltration
Comorbidities (diabetes, heart disease, chronic lung disease, tuberculosis, human
immunodeficiency virus, severe liver disease, severe kidney impairment)
If female, known to be pregnant
Second randomisation
In addition to the above:
Type of ventilation support currently required (none, CPAP alone, non-invasive
ventilation, high-flow nasal oxygen, mechanical ventilation, ECMO)
Latest oxygen saturation measurement (%)
Latest CRP measurement (mg/L)
Latest ferritin measurement (ng/mL)
Latest creatinine measurement (µmol/L)
Allocation in first randomisation
Interval between first and second randomisation
The number and percentage will be presented for binary and categorical variables. The mean
and standard deviation or the median and the interquartile range will be presented for
continuous variables, or the range if appropriate. There will be no tests of statistical
significance performed nor confidence intervals calculated for differences between
randomised groups on any baseline variable.
4.3 Completeness of follow-up
All reasonable efforts will be taken to minimise loss to follow-up, which is expected to be
minimal as data collection for primary and secondary outcomes using trial-specific eCRFs is
combined with linkage to routine clinical data on study outcomes from NHS Digital, ICNARC,
and similar organisations in the devolved nations.
The number and percentage of participants with follow-up information at day 28 and at 6
months after the main randomisation will be reported. Data will be shown for each of the
following: all-cause mortality, hospital discharge status, ventilation status, and will be shown
for each randomised group for the main and second randomisation separately.
4.4 Adherence to treatment
The number and proportion of patients who did not receive the treatment they were
allocated to will be reported. If any other trial treatment options were known to be received,
instead of or in addition to, the allocated treatment during the 28 day follow-up period after

RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 14 of 20
the first randomisation, these will be collected and reported. Details on the number of days
(or doses) of treatment received will be reported for all trial treatments received where
available.
5 COMPARATIVE ANALYSES
For all outcomes, the primary analysis will be performed on the intention to treat (ITT)
population at 28 days after the main randomisation. An ITT analysis of all outcomes at 6
months post-randomisation will also be conducted.
Pairwise comparisons will be made between each treatment arm and the no additional
treatment arm (reference group) in that particular randomisation (main randomisation part
A, main randomisation part B, and second randomisation). Since not all treatments may be
available or suitable for all patients, those in the no additional treatment arm will only be
included in a given comparison if, at the point of their randomisation, they could alternatively
have been randomised to the active treatment of interest (i.e. the active treatment was
available at the time and it was not contra-indicated). The same applies to treatment arms
added at a later stage; they will only be compared to those patients recruited concurrently.
5.1 Main randomisation part A
Primary outcome
Mortality (all-cause) will be summarised with counts and percentages by randomised
comparison group. A time-to-event analysis will be conducted using the log-rank test, with
the p-value reported. Kaplan-Meier estimates for the time to event will also be plotted (with
associated log-rank p-values). The log-rank ‘observed minus expected’ statistic (and its
variance) will be used to estimate the average event rate ratio and confidence interval for
each treatment group versus the no additional treatment group.
5
For the primary outcome,
discharge alive before the relevant time period (28 days) will be assumed as absence of the
event (unless there is additional data confirming otherwise).
Secondary outcomes
5.1.2.1 Time to discharge from hospital
A time-to-event analysis will be used to compare each treatment group with the no additional
treatment group using Kaplan-Meier and the log-rank test, as described above. Patients who
die in hospital will be censored after 28 days. This gives an unbiased estimate of the recovery
rate and comparable estimates to the competing risks approach in the absence of other
censoring (which is expected to be very minimal).
6
5.1.2.2 Use of mechanical ventilation/ECMO or death (among those not on ventilation or
ECMO at randomisation)
Counts and percentages will be presented by randomised group and the risk ratio will be
calculated for each pairwise comparison with the no additional treatment arm, with
confidence intervals and p-values reported. The absolute risk difference will also be presented
with confidence intervals. Patients who were already on ventilation at randomisation will be
excluded from the denominator.
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 15 of 20
Subsidiary clinical outcomes
5.1.3.1 Cause-specific mortality
Cause-specific mortality will be analysed in a similar manner to the primary outcome. Deaths
from other causes will be censored at the date of death and a separate survival curve will be
presented for each cause of death (COVID-19, other infection, cardiovascular and other).
5.1.3.2 Use of renal dialysis or haemofiltration
Counts and percentages will be presented by randomised group and the risk ratio will be
calculated for each pairwise comparison with the no additional treatment arm, with
confidence intervals and p-values reported. The absolute risk difference will also be presented
with confidence intervals. Patients who were already on renal dialysis or haemofiltration at
randomisation will be excluded from the denominator.
5.1.3.3 Major cardiac arrhythmia
Counts and percentages will be presented by randomised group and the risk ratio for any
major cardiac arrhythmia will be calculated for each pairwise comparison with the no
additional treatment arm, with confidence intervals and p-values reported. The absolute risk
difference will also be presented with confidence intervals. Type of arrhythmia will also be
described: (i) atrial flutter or fibrillation; (ii) supraventricular tachycardia; (iii) ventricular
tachycardia; (iv) ventricular fibrillation; (v) atrioventricular block requiring intervention, with
subtotals for (i)-(ii) and (iii)-(iv).
5.1.3.4 Use of ventilation (overall and by type)
Counts and percentages will be presented by randomised group for patients who received
any assisted ventilation. Patients who were already on assisted ventilation at randomisation
will be excluded from the denominator. The number of patients receiving the different types
of ventilation will also be reported: (i) CPAP; (ii) other non-invasive; (iii) high-flow nasal
oxygen; (iv) mechanical; (v) ECMO, with subtotals for (i)-(iii) (non-invasive) and (iv)-(v)
(invasive).
5.1.3.5 Duration of ventilation (overall and by type)
The mean (SD) duration of ventilation will be calculated in days from the main randomisation
for each randomised group in those who received ventilation, separately for survivors and
non-survivors. This will be reported overall for any assisted ventilation and separately for
mechanical ventilation or ECMO. The mean difference and confidence intervals will be
presented for each pairwise comparison with the no additional treatment arm.
5.2 Main randomisation part B
For the evaluation of treatment effect in the factorial design, the main effect of convalescent
plasma across all arms in main randomisation part A combined, will be presented and tested,
as described in 5.1. Data stratified by allocation in part A will also be reported to aid
interpretation, but no tests for statistical interaction will be performed.
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 16 of 20
Additional safety data will be collected in a subset of patients randomised to part B. These
will be tabulated separately by allocation (convalescent plasma versus no additional
treatment): (i) sudden worsening in respiratory status; (ii) severe allergic reaction; (iii)
temperature >39°C or ≥2°C rise since randomisation; (iv) sudden hypotension, clinical
haemolysis and thrombotic event.
5.3 Second randomisation
Evaluation of treatment effects in the main randomisation and the second randomisation will
be conducted independently, as described in 5.1. In addition to the overall comparison for
Tocilizumab vs no additional treatment, results will be stratified according to allocation in the
main randomisation (part A and part B), however no interaction tests will be performed
between the allocations in the two stages.
5.4 Pre-specified subgroup analyses
Pre-specified subgroup analyses will be conducted for the main randomisation (part A and
part B) and the second randomisation, for the following outcomes:
Mortality (all-cause)
Time to discharge from hospital
Use of mechanical ventilation/ECMO or death
The analyses will be conducted using a test for heterogeneity (or test for trend for 3 or more
ordered groups). Results will be presented on forest plots as event rate ratios (or risk ratios)
with confidence intervals. The following subgroups will be examined:
Risk group (three risk groups with approximately equal number of deaths based on
factors recorded at randomisation)
Requirement for respiratory support at randomisation (None; Oxygen only;
Ventilation or ECMO)
Time since illness onset (≤7 days; >7 days)
Age (<70; 70-79; 80+ years)
Sex (Male; Female)
Ethnicity (White; Black, Asian or Minority Ethnic; Unknown)
Additional analyses will set the results for children (<18 years) and pregnant women in the
context of the overall results.
5.5 Significance levels and adjustment of p-values for multiplicity
Evaluation of the primary trial (main randomisation) and secondary randomisation will be
conducted independently and no adjustment be made for these. Formal adjustment will not
be made for multiple treatment comparisons, the testing of secondary and subsidiary
outcomes, or subgroup analyses. 95% confidence intervals will be presented for estimates of
between-group effects throughout.
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 17 of 20
5.6 Statistical software employed
The statistical software SAS version 9.4, R Studio 3.6.2 and Stata/SE version 15 (or later) for
Windows will be used for the interim and final analyses.
5.7 Data standards and coding terminology
Datasets for analysis will be prepared using CDISC standards for SDTM and ADaM. Wherever
possible, clinical outcomes (which may be obtained in a variety of standards, including ICD10
and OPCS-4) will be coded using MedDRA version 20.1.
6 SAFETY DATA
Suspected serious adverse reactions (SSARs) and suspected unexpected serious adverse
reactions (SUSARs) will be listed by trial allocation.
7 ADDITIONAL EXPLORATORY ANALYSIS
Any post-hoc analysis requested by the oversight committees, a journal editor or referees will
be labelled explicitly as such. Any further future analyses not specified in the analysis protocol
will be exploratory in nature and will be documented in a separate statistical analysis plan.
8 DIFFERENCES FROM PROTOCOL V6.0
Use and duration of ventilation are described as secondary objectives in the protocol, and
listed as subsidiary outcomes in the statistical analysis plan. The testing of multiple treatment
arms will not formally be adjusted for, but given the number of comparisons, due allowance
will be made in their interpretation. Formal methods of adjustment for multiplicity were not
adopted because of treatment arms being added over time (including the factorial
convalescent plasma comparison), unequal recruitment into each arm, and the ultimate
number of treatments under evaluation not known in advance. While methods for these
situations exist it was felt that the resulting change in level of significance was not
appropriate.
9 REFERENCES
9.1 Trial documents
Dummy tables and the data derivation document can be found in the RECOVERY trial
directory and will be published with this SAP on the trial website.
9.2 Other references
1. Gamble C, Krishan A, Stocken D, Lewis S, Juszczak E, Doré C, Williamson PR, Altman
DG, Montgomery A, Lim P, Berlin J, Senn S, Day S, Barbachano Y, Loder E. Guidelines for the
Content of Statistical Analysis Plans in Clinical Trials. JAMA 2017;318(23):2337-2343.
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 18 of 20
2. Schulz KF, Altman DG, Moher D for the CONSORT Group. CONSORT 2010 Statement:
updated guidelines for reporting parallel group randomised trials. BMJ 2010;340:698-702.
3. Juszczak E, Altman DG, Hopewell S, Schulz KF. Reporting of multi-arm parallel-group
randomized trials: extension of the CONSORT 2010 statement. JAMA 2019;321(16):1610-
1620.
4. Dimairo M, Coates E, Pallmann P, Todd S, Julious SA, Jaki T, Wason J, Mander AP,
Weir CJ, Koenig F, Walton MK. Development process of a consensus-driven CONSORT
extension for randomised trials using an adaptive design. BMC medicine 2018;16(1):210.
5. Peto R, Pike MC, Armitage P, Breslow NE, Cox DR, Howard SV, Mantel N, McPherson
K, Peto J, Smith PG. Design and analysis of randomized clinical trials requiring prolonged
observation of each patient. Part II: analysis and examples. Br J Cancer 1977;35:1-39.
6. Betensky RA and Schoenfeld DA. Nonparametric Estimation in a Cure Model with
Random Cure Times. Biometrics 2001;57:282-286.
RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 19 of 20
10 APPROVAL
Lead Trial
Statistician
Name: Dr Louise Linsell
Signature:
Date:
Chief
Investigator
Name: Professor Peter Horby
Signature:
Date:
Deputy Chief
Investigator
Name: Professor Martin Landray
Signature:
Date:
Steering
Committee
Statistician
Name: Associate Professor Ed Juszczak
Signature:
Date:
Steering
Committee
Statistician
Name: Professor Alan Montgomery
Signature:
Date:
Steering
Committee
Statistician
Name: Professor Thomas Jaki
Signature:
Date:

RECOVERY SAP Version date: 09 June 2020 Version number: 1.0
RECOVERY SAP v1.0 09_06_20.DOCX Page 20 of 20
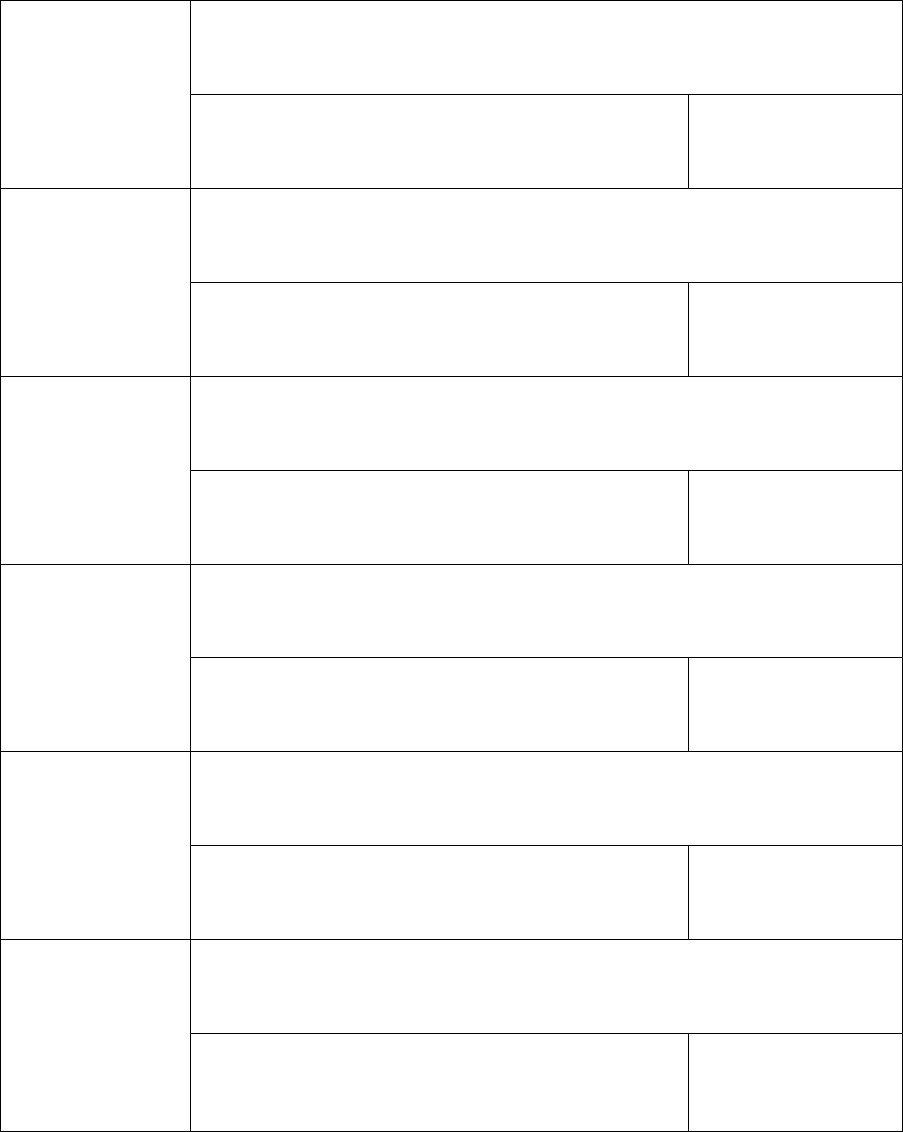
11 DOCUMENT HISTORY
Version
Date
Edited
by
Comments/Justification
Timing in
relation to
unblinded
interim
monitoring
Timing in
relation to
unblinding of
Trial
Statisticians
0.1
20/03/20
LL/JB
First draft.
Prior
Prior
0.2
01/04/20
LL/JB
Comments and amendments
from Martin Landray, Jonathan
Emberson & Natalie Staplin.
Also aligned with updated
protocol and CRFs.
Prior
Prior
0.3
01/04/20
EJ/LL
Further edits and comments.
Prior
Prior
0.4
07/04/20
JB/EJ/LL
Following statistics group
meeting on 02/04/20.
Prior
Prior
0.5
22/04/20
JB/LL/EJ
Following statistics group
meeting on 09/04/20 and
further protocol update.
After
Prior
0.6
24/04/20
LL
Following statistics group
meeting on 23/04/20.
After
Prior
0.7
10/05/20
LL
Protocol update.
After
Prior
0.8
15/05/20
LL
Following statistics group
meeting on 15/05/20.
After
Prior
0.9
27/05/20
LL
Further comments from SC
members prior to interim
analysis on 28/05/20.
After
Prior
1.0
09/06/20
LL
Revised following the stopping
of the hydrochloroquine arm,
and prior to the trial statisticians
receiving unblinded data for this
arm.
After
Prior
